SBIR/STTR Needs TLC PDQ!
You can't fertilize America's Seed Fund with bulls**t
I’ve moved the usual frontmatter for NIH/HHS posts to the end so that we can get right into the topic for this post. Also, I think you probably know the drill by now. The usual lead-in is now down at the bottom under the Explainer and Disclaimer header.
Today’s post is about the Small Business Innovation Research (SBIR) program and the related Small Business Technology Transfer (STTR) program. This is a Federal program that originated in 1982 with the Small Business Innovation Development Act and is run and overseen by the Small Business Administration, an independent agency of the United States Government. The purpose of the SBIR/STTR program is to allocate public funds to invest in the growth of small businesses, particularly small businesses breaking new and innovative ground in scientific and technological areas. The goal is to create paths to commercialization for innovations supported by Federal research and development dollars. Importantly, funds from the SBIR and STTR programs do not dilute the equity of any of the nearly 4,000 businesses that participate annually in the program (see https://www.sbir.gov/ for more details). Unlike private capital or private equity interests that purchase a portion of the company expecting a direct financial return on investment (ROI), the SBIR/STTR program’s desired ROI is merely that the supported businesses succeed and contribute to the whole country’s economic prosperity.
Called “America’s Seed Fund” the SBIR/STTR program has had a huge positive impact on the United States’ capacity for ensuring that Federally supported scientific and technological breakthroughs have a path all the way to success in the global marketplace, with all of the job creation and economic activity that comes with that. But, there have been some recent changes to the SBIR/STTR program.
Here’s the high-level summary:
There is currently no SBIR/STTR program in the United States.
That’s it. That’s the summary.
Okay, there’s a bit more to it. Like many Federal programs and even agencies, the SBIR/STTR program must be authorized by Congress to even exist. Note that authorization is technically separate from appropriation, even though both sometimes happen together in the same bill. Authorization brings the thing into being. Appropriation gives it a financial blood supply. (For example, the authorizations that allow/require NIH to exist and do stuff are different from the annual appropriations process that we talk about when we talk about the “NIH budget” that will fund those activities. You can read more deeply at this link if you’re interested: https://www.congress.gov/crs-product/R41705 )
As you might imagine, when Congress legislates the authority to do stuff, they might not want that authority to exist permanently without at least periodic re-examination. And so, programs and agencies typically require periodic reauthorization. For example, you might’ve heard about “puhDOOFuh”, otherwise known as the Prescription Drug User Fee Act (PDUFA). This is the law that authorizes the FDA to do a whole bunch of core things that we expect the FDA to do. We’re on PDUFA VII which runs through fiscal year 2027 (remember the Federal fiscal year ends on September 30, so FY 2027 begins on October 1, 2026 because of course it does), at which point it’ll need to be reauthorized.
The SBIR/STTR program is no different. It requires periodic reauthorization. The big problem is that the program’s authorization expired on September 30, 2025 (the end of FY 2025), and as of January 6, 2026 the program has not been reauthorized. Very simply stated, that means that currently the United States does not have an SBIR/STTR program. Practically, this means that existing awards can continue (provided funds are available), but no new awards can be issued and no new solicitations/funding opportunities under the SBIR/STTR program can be issued. Remember that for NIH grants including SBIR/STTR grants, the noncompeting project years must still be awarded anew each year. So, if you have a 3 year project supported by an NIH SBIR/STTR grant and you’re finishing year 1, year 2 is not considered an “existing award” because each project year is a separate award.
There’s one other slightly wonky point that’s really important to note because there are big implications. One might think, “Why can’t NIH Institutes and Centers just fund SBIR/STTR awards with other funds?” Good question, and the answer is that it’s forbidden/illegal. This is a bigger problem than it first appears. It means that there’s no way to backstop the SBIR/STTR program until Congress reauthorizes it. But it also means that any ongoing SBIR/STTR activities that involve human participants – y’know, stuff like the clinical trials that support the products and services developed by the NIH SBIR/STTR grantees – have absolutely no way of being supported until the program is reauthorized, not even if participant safety is at risk from abrupt cessation of activities that are part of an affected SBIR/STTR award. So, imagine a trial of a medical device that’s implanted and then retrieved. If the trial is supported by an SBIR/STTR grant that now has no way for the subsequent years to be funded, what happens to those participants?
Yeah.
How Big Is the Problem?
Pretty big.
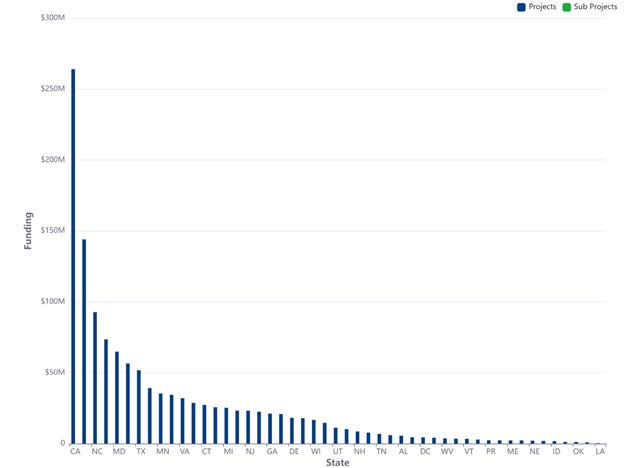
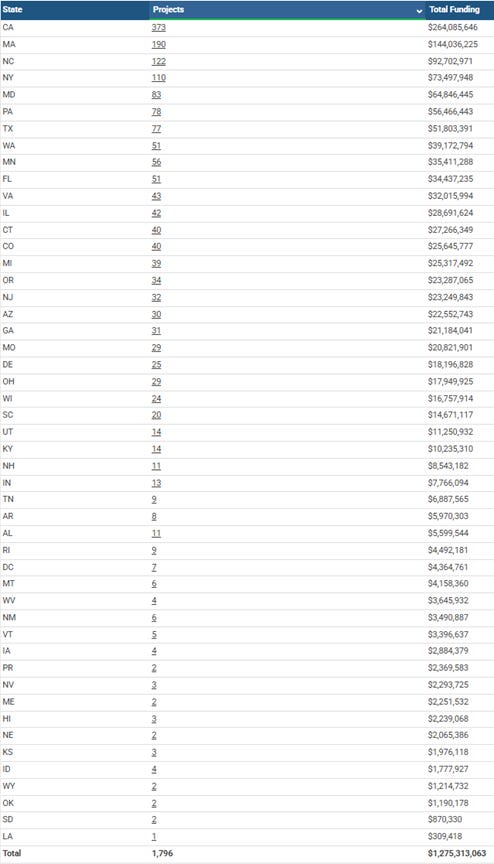
First, let’s look at what NIH SBIR/STTR funding looked like in FY 2025, which encompasses the year right up to the expiration of the program’s authorization. Here’s the plot of funding by state for FY 2025:
And here’s the breakdown of the numbers by state:
Here’s the search I did in NIH RePORTER: https://reporter.nih.gov/search/q8OPvluDnUmiTHQqJ--CCw/projects/charts?shared=true
The data for FY 2024 are similar though even more impressive, with the NIH SBIR/STTR program supporting 2,016 projects at a total funding level of $1,326,392,388. You can see those data from this search: https://reporter.nih.gov/search/2E-d9oJi1UiVAHROJJmuYg/projects/charts?shared=true (note that when you follow these links, you’ll probably have to change the x-axis and y-axis on the charts to get “Funding” on the y-axis and “State” on the x-axis)
For comparison and context, let’s look at a state like Florida and what the state’s investment in this kind of small business development looks like. The comparison is only a rough one at best, for several reasons. The 2025 fiscal year for the state of FL (July 1, 2025 – June 30, 2026) is different than the Federal fiscal year 2025 (October 1, 2024 – September 30, 2025). Florida’s investment in small businesses is reported as a combination of positive investments (e.g., grants, infrastructure construction), loan programs, and tax cuts. Since NIH cannot do anything about taxes and does not issue loans, I’ll try to limit comparisons only to non-dilutive positive investments, excluding FL’s small business loans (which of course must be paid back to the state) and tax cuts/breaks (which do affect a business’s balance sheet and do incentivize business development but do not directly support R&D or commercialization). Looking at the programs listed on the website* https://www.flgov.com/eog/news/press/2025/governor-ron-desantis-signs-florida-fiscal-year-2025-2026-budget Florida’s most recent state budget commits $50M to the Job Growth Grant Fund, $22M to the Rural Infrastructure Fund, and $2M to assist veterans developing a small business. That’s a total of $74M, which is definitely not couch cushion money.
Here’s the thing. FROM NIH’S SBIR/STTR PROGRAM ALONE, Florida *could* be receiving almost $35M in additional small business development support. Not from loans, or from private equity centered outside the state, or from tax cuts, or from anything other than a +$35M on the balance sheet coming from Federal dollars to support the commercialization of biomedical innovations in the state of Florida. And that’s an almost 50% increase in small business support coming JUST FROM NIH! That doesn’t even consider SBIR/STTR funding that might come to FL from the other 10 Federal agencies that participate in the program when it exists (and those can be big dollars - e.g., the Dept of Defense’s SBIR/STTR activities far exceed those of NIH). And, of course, none of this takes into account the future economic growth that could have come from SBIR/STTR investment in small businesses – nationally, this means on average over 65,000 jobs annually and billions of dollars of additional economic activity annually.
That’s what’s missing right now, across the whole United States. Missing at a time when the job market is pretty not great (unemployment in Nov 2025 sitting at 4.6%, representing a steady increasing trend over 2025 and the highest November unemployment level since 2020 - see https://data.bls.gov/timeseries/LNS14000000 for details). Missing at a time when the innovation economy should be booming the U.S., but instead the U.S. is flat while other nations are making big moves to lead the global innovation economy (see https://www.wipo.int/web-publications/global-innovation-index-2025/en/gii-2025-results.html for more info). Investment in high tech and biotech is going full steam ahead in other competitor nations, and they see an opportunity to eat our lunch. The ROI for the SBIR/STTR program is so clear. What’s not clear is why the U.S. Congress can’t seem to see it’s way to say, “Keep doing what’s been working well for the last 43 years.”
What Can We Do?
1. Keep the pressure on elected officials. This is a problem that can only be solved by legislative action, and the benefit to every state for reauthorization of the SBIR/STTR program seems immediately obvious. Call your Representatives and Senators. Let them know that you insist they reauthorize the SBIR/STTR program with all due haste, and that their record of support for job creation via supporting small business development will definitely be a factor in how you vote in future elections. If you live in a state with a Senator serving on the Senate Small Business and Entrepreneurship Committee, you *definitely* need to let them hear you – members can be found here: https://www.sbc.senate.gov/public/ This is not an obscure kind of thing that only DC wonks care about. The effects are very local.
2. Pay close attention to those who are following this issue. The broad strokes are pretty easy to grasp, but the devil is always in the details. I’ve listed some of the sources I consulted to write this in the Further Reading section below, and there are plenty of others. Seek them out and stay alert.
3. Plan ahead and expect frustrations. Even if the SBIR/STTR program were reauthorized today, it’d be a bumpy ride. There’s a big backlog of applications to be considered for funding and to be reviewed. At NIH, how funding decisions are being made is in flux, and we’re going to have to live our way to understanding what that means for SBIR/STTR grants (and all the other funding mechanisms, for that matter). And small businesses typically have to make decisions and take action on timeframes that are *much* shorter than what NIH and other government agencies operate on. Think ahead, plan ahead, and bring your buckets full of patience and grace.
4. Be strategic now. It is likely that the SBIR/STTR program will be reauthorized. I don’t know when, or whether it’ll look different than it has previously. Some of that is yet to be determined (see point #1 above). But don’t wait to plan. Think about what you have in the works that might be a good fit for SBIR/STTR support. Take a good look at what it would take to put an application together – person hours, partnerships, juice-to-squeeze ratio (i.e., would the work required to prep and submit the application and the work you’d be committed to doing if funded be worth the likely award amount?). Game out the scenarios. Build the partnerships ahead of time. TALK TO PROGRAM OFFICERS! Ask them where things are right now and where things are headed. Ask them about alternative funding mechanisms that might accomplish some of the same goals as SBIR/STTR funding, programs such as the NIH Blueprint MedTech program or NHLBI’s Catalyze program. Be ready to move when the time is right.
5. Remember the importance of stories. People aren’t moved by numbers, they’re moved by the stories behind the numbers. When engaging with your elected officials, with friends and family, with institutional leaders, with journalists, with anyone, remember to tell your compelling story. The group 27 UNIHTED (https://www.27unihted.org/) is collecting impact stories about the SBIR/STTR program - https://www.27unihted.org/sbir-story More of this, please!
Some programs are difficult to understand in terms of why investment of Federal dollars makes sense. In my opinion, the SBIR/STTR program is definitely not one that’s hard to understand. Innovation good. Business good. Economic growth good. Unemployment bad.
Further Reading
https://www.sbc.senate.gov/public/
https://www.wipo.int/web-publications/global-innovation-index-2025/en/gii-2025-results.html
https://www.congress.gov/crs-product/R41705
https://www.sbir.gov/ and https://www.sbir.gov/impact/impact-reports and https://www.sbir.gov/success-stories
https://www.ebhoward.com/as-of-january-2026-where-things-stand-with-sbir-sttr-reauthorization/
*Weirdly, the State of Florida’s main website is https://www.myflorida.gov/ but the link to the Executive Office of the Governor is not a .gov website but rather directs to the flgov.com domain. Hmmm… not sure what that means, but there we are.
Explainer and Disclaimer
The purpose of this post is to try to bring a teeny bit of clarity to the roiling landscape of biomedical research, healthcare, and public health in the U.S. This isn’t about outrage or blame or anything like that. I’m just hoping to highlight some things that I think are important to know about in the present moment, offer my thoughts on why they’re important to know about, and offer some ideas on what people and institutions do with the information. I’ll focus on things related to the NIH and the Dept of Health and Human Services, though if there are goings-on in other sectors that are likely to have a big impact on biomedical research and public health, I’ll go there.
SUPER DUPER IMPORTANT CAVEAT: Though I am a former federal employee and will look at things through that lens, I am not currently a federal employee. The golden rule is always this: TALK TO YOUR POINT OF CONTACT – your program officer, contracting official, whomever is appropriate. Talk to them early and as often as it takes. And if you’re not sure who that is, press pause right now and go do some digging. When appropriate here, I’ll offer thoughts on who the right POC might be for a specific thing. But there’s no substitute for up-to-date information coming straight from the people tasked with implementing policies, EOs, random tweets, whatever.